CLASSIFICATION OF MATTER
Chemists study the structures, physical properties, and chemical properties of material substances. These consist of matter, which is anything that occupies space and has mass. Gold and iridium are matter, as are peanuts, people, and postage stamps. Smoke, smog, and laughing gas are matter. Energy, light, and sound, however, are not matter; ideas and emotions are also not matter.
The mass of an object is the quantity of matter it contains. Do not confuse an object’s mass with itsweight, which is a force caused by the gravitational attraction that operates on the object. Mass is a fundamental property of an object that does not depend on its location.In physical terms, the mass of an object is directly proportional to the force required to change its speed or direction. A more detailed discussion of the differences between weight and mass and the units used to measure them is included in Essential Skills 1 (Section 1.9). Weight, on the other hand, depends on the location of an object. An astronaut whose mass is 95 kg weighs about 210 lb on Earth but only about 35 lb on the moon because the gravitational force he or she experiences on the moon is approximately one-sixth the force experienced on Earth. For practical purposes, weight and mass are often used interchangeably in laboratories. Because the force of gravity is considered to be the same everywhere on Earth’s surface, 2.2 lb (a weight) equals 1.0 kg (a mass), regardless of the location of the laboratory on Earth.
Under normal conditions, there are three distinct states of matter: solids, liquids, and gases. Solids are relatively rigid and have fixed shapes and volumes. A rock, for example, is a solid. In contrast, liquids have fixed volumes but flow to assume the shape of their containers, such as a beverage in a can. Gases, such as air in an automobile tire, have neither fixed shapes nor fixed volumes and expand to completely fill their containers. Whereas the volume of gases strongly depends on their temperature and pressure (the amount of force exerted on a given area), the volumes of liquids and solids are virtually independent of temperature and pressure. Matter can often change from one physical state to another in a process called a physical change. For example, liquid water can be heated to form a gas called steam, or steam can be cooled to form liquid water. However, such changes of state do not affect the chemical composition of the substance.
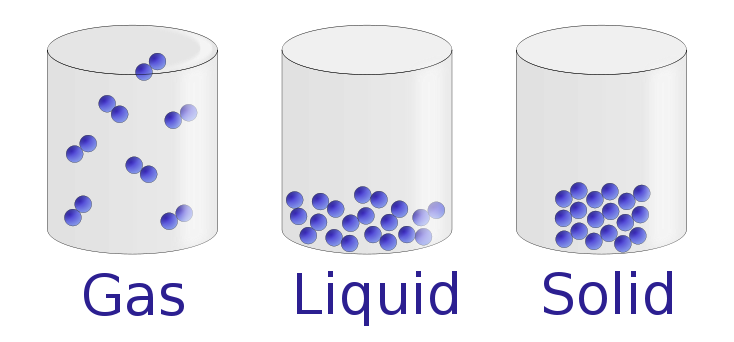
Figure : The Three States of Matter. Solids have a defined shape and volume. Liquids have a fixed volume but flow to assume the shape of their containers. Gases completely fill their containers, regardless of volume. Figure used with permission from Wikipedia
Pure Substances and Mixtures
A pure chemical substance is any matter that has a fixed chemical composition and characteristic properties. Oxygen, for example, is a pure chemical substance that is a colorless, odorless gas at 25°C. Very few samples of matter consist of pure substances; instead, most are mixtures, which are combinations of two or more pure substances in variable proportions in which the individual substances retain their identity. Air, tap water, milk, blue cheese, bread, and dirt are all mixtures. If all portions of a material are in the same state, have no visible boundaries, and are uniform throughout, then the material is homogeneous. Examples of homogeneous mixtures are the air we breathe and the tap water we drink. Homogeneous mixtures are also called solutions. Thus air is a solution of nitrogen, oxygen, water vapor, carbon dioxide, and several other gases; tap water is a solution of small amounts of several substances in water. The specific compositions of both of these solutions are not fixed, however, but depend on both source and location; for example, the composition of tap water in Boise, Idaho, is not the same as the composition of tap water in Buffalo, New York. Although most solutions we encounter are liquid, solutions can also be solid. The gray substance still used by some dentists to fill tooth cavities is a complex solid solution that contains 50% mercury and 50% of a powder that contains mostly silver, tin, and copper, with small amounts of zinc and mercury. Solid solutions of two or more metals are commonly called alloys.
If the composition of a material is not completely uniform, then it is heterogeneous (e.g., chocolate chip cookie dough, blue cheese, and dirt). Mixtures that appear to be homogeneous are often found to be heterogeneous after microscopic examination. Milk, for example, appears to be homogeneous, but when examined under a microscope, it clearly consists of tiny globules of fat and protein dispersed in water. The components of heterogeneous mixtures can usually be separated by simple means. Solid-liquid mixtures such as sand in water or tea leaves in tea are readily separated by filtration, which consists of passing the mixture through a barrier, such as a strainer, with holes or pores that are smaller than the solid particles. In principle, mixtures of two or more solids, such as sugar and salt, can be separated by microscopic inspection and sorting. More complex operations are usually necessary, though, such as when separating gold nuggets from river gravel by panning. First solid material is filtered from river water; then the solids are separated by inspection. If gold is embedded in rock, it may have to be isolated using chemical methods.
-and-Milk-(left).jpg?revision=1)
Figure : A Heterogeneous Mixture. Under a microscope, whole milk is actually a heterogeneous mixture composed of globules of fat and protein dispersed in water. Figure used with permission from Wikipedia
Homogeneous mixtures (solutions) can be separated into their component substances by physical processes that rely on differences in some physical property, such as differences in their boiling points. Two of these separation methods are distillation and crystallization. Distillation makes use of differences in volatility, a measure of how easily a substance is converted to a gas at a given temperature. A simple distillation apparatus for separating a mixture of substances, at least one of which is a liquid. The most volatile component boils first and is condensed back to a liquid in the water-cooled condenser, from which it flows into the receiving flask. If a solution of salt and water is distilled, for example, the more volatile component, pure water, collects in the receiving flask, while the salt remains in the distillation flask.


Please explain the difference gas, liquid, and solid?
BalasHapusSolids: have a shape with a certain volume, the distance between the particles tends to be very tight and the particles can not move freely but only vibrate or spin in place alone, the force of attraction between molecules is very strong.
Hapus- Liquid: has a certain but not definite volume determined by the occupied medium, has a surface that is always flat, the distance between the particles slightly tenuous with the particles can move freely but limited, the attraction between the molecules is still proven still Difficult to unravel.
- Gas substances: have no fixed or cheap volumes and shapes change, the distance between particles is very tenuous with the particles can move freely, the pull force between molecules almost nothing, can fill all the existing space.
Can gass be solid?
BalasHapusYes, gass can be solid.
HapusAs we know that milk is natural and some are artificial, there are also artificial sweetened condensed milk and some milk powder, which I want to ask which milk is homogeneous and which milk is heterogeneous?
BalasHapusHomogenised milk canned milk, soy milk milk estrogen.
HapusHow to separate the gas mixture?
BalasHapusFiltering or better known as filtration
HapusIn a Filtering process done based on the difference in particle size. In the filtration process use a filter example such as filter paper, so that very small particles can pass through the filter. The filtrate result in the particles is called the filtrate, while the larger particles and retained on the filter paper are in contact with the residue.
2. Distillation (distillation)
Distillation is a technique in a mixed separation that occurs based on the difference of a boiling point comprising each component present in a mixture. The process of separation of the mixture is done by way of distillation is done in two ways, the first is by evaporation condensation. The mixture is first heated to above the boiling point of the substance to be separated. Since the boiling point of the substance to be separated is lower than the boiling point of the mixture, the substance will evaporate first. The vapor is then cooled to become a liquid.
BalasHapusIs there a theory about atoms?
Atomic Theory of John Dalton
HapusIn 1803, John Dalton expressed his opinion on the atom. Dalton's atomic theory is based on two laws, namely mass conservation law (Lavoisier law) and fixed order law (law of prouts). Lavosier states that "The total mass of substances before the reaction will always be equal to the total mass of the reaction products." While Prouts states that "Comparison of the mass of the elements in a compound is always fixed". Of these two laws Dalton expressed his opinion about atoms as follows:
1. Atom is the smallest part of matter that can not be divided again
2. Atoms are described as very small solid spheres, an element having atoms identical and different for different elements
3. Atoms combine to form compounds with simple and integer ratios. For example water consists of hydrogen atoms and oxygen atoms.
Specify the elemen form? Try to explain and the difference?
BalasHapusplease give example of desgtilation in chemical reaction, i mean the reaction
BalasHapus